UNIT 7: Matter(6 DAYS)
Gases, liquids, and especially solids surround us and give form to our world. Chemistry at its most fundamental level is about atoms and the forces that act between them to form larger structural units. But the matter that we experience with our senses is far removed from this level. This unit will help you see how these macroscopic properties of matter depend on the microscopic particles of which it is composed. |
NC Standards
PSc 2.1.1 Classify matter as: homogenous or heterogenous; pure substance or mixture; element or compound; metals, nonmetals or metalliods; solution, colloid or suspension.
PSc 2.1.2 Explain the phases of matter and the physical changes that matter undergoes
PSc 2.1.3 Compare physical and chemical propertie of various types of matter
PSc 2.1.4 Interpret data presented in Bohr model diagrams and dot diagrams for atoms and ions of elements 1-18
PSc 2.1.2 Explain the phases of matter and the physical changes that matter undergoes
PSc 2.1.3 Compare physical and chemical propertie of various types of matter
PSc 2.1.4 Interpret data presented in Bohr model diagrams and dot diagrams for atoms and ions of elements 1-18
Essential Questions
7.1 States of MatterDAY 1: TYPES OF MATTER
HOMEWORK: CLASSIFYING MATTER on quia
DAY 2: CLASSIFYING MATTER
Volume LAB
7.2 Physical & Chemical ChangesDAY 3: PHYSICAL/CHEMICAL CHANGES / PHASE CHANGES
|
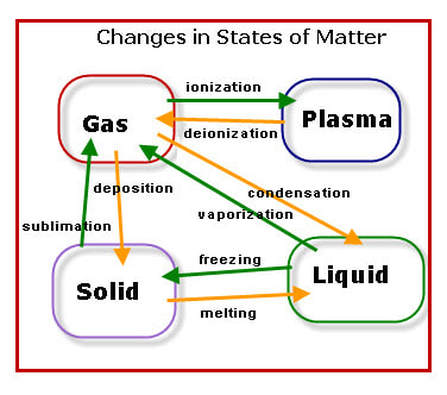
Phase Change Diagram Video
| ||||||||||||||||||||||||||
DAY 4: . HEATING & COOLING CURVES
| changes_of_state_ppt5.ppt |
| changes_of_state_ppt_notes_blanks.docx |
| phases_of_matter_worksheet.docx |
DAY 6: UNIT TEST
LABS
Separation of a Mixture Oobleck Lab
Volume Lab Classifying Solution Mixtures
Volume Lab Classifying Solution Mixtures